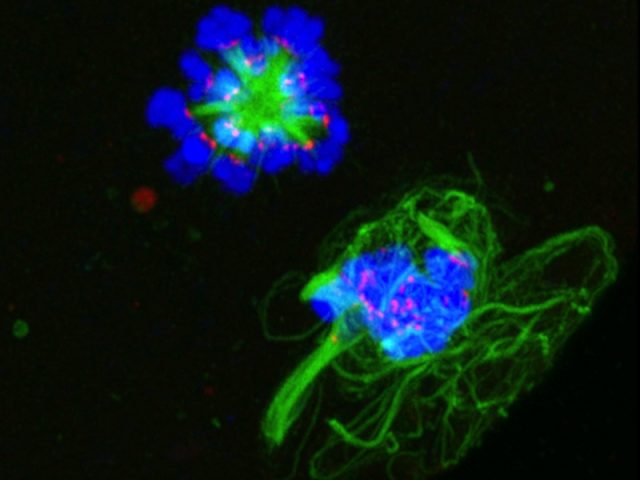
Ovarian determinants
The primary goals of the laboratory are to understand the development of the ovarian follicle, identify markers and determinants of egg quality, and discover how this basic biology can be applied to patients. This research includes the biological, cellular, and molecular mechanisms that regulate the development of the ovarian follicle, such as the study of endocrine, structural, and other environmental cues in the ovary, and…
The Woodruff Lab aims to develop noninvasive ways to determine when a follicle has reached full maturity in vitro. The team uses a variety of model systems to investigate such strategies, including mouse and bovine. They are developing integrated methods to monitor follicle maturity involving noninvasive measurements of follicle diameter, hormone production, secreted proteins, chromosome dynamics, and chemical signatures. Through such individualized follicle monitoring, the lab will ultimately establish a culture system that best recapitulates in…
Patient Translation
The Woodruff Lab applies the knowledge of follicle development and markers of egg quality to improve the quality of life for young women with diseases, such as cancer. Cancer treatments including chemotherapy, radiation, or surgery may impair the later reproductive ability of survivors. We aim to prevent this damage by developing new in vitro methods to foster follicle development.
In vitro follicle growth
A major goal in the lab is to develop methods to grow…