Like most biological research in the United States, the Woodruff Lab receives the majority of funding from the American taxpayer via the National Institutes of Health. Some of the Woodruff Lab's current, proposed and completed funding comes from the following grants:
Assisted Reproductive Technologies (ART) have resulted in the birth of millions of individuals worldwide, however, this technology still has many imperfections including low efficiencywith a live birth rate of ~34-40%, and high risk of multiple gestation with its myriad associated risks. Therefore, a reliable biomarker to predict the embryo with the highest developmental potential has been badly needed to advance this technology. Activation or fertilization of mammalian eggs initiates a series of extracellular “zinc (Zn) sparks” that are highly coordinated with intracellular calcium (Ca2+) transients and also necessary to induce the egg-to-embryo transition. Although Ca2+ transients are known to correlate with embryo development, they cannot serve as clinical biomarkers as they occur intracellularly. In contrast, because Zn is released into the extracellular space, this element can be detected in a non-invasive manner and objectively quantified. These characteristics make Zn sparks an ideal biomarker of embryo quality. However, despite the prominence of this Zn-efflux event, it is unknown if the Zn sparks have any effects on the embryonic developmental outcomes. We therefore hypothesize that profiles of Zn sparks are correlated with embryo development and can be used as a biomarker of embryo quality.
One of the most compelling unanswered questions in human biology is how just one out of half a million oocytes is selected each month to complete its development and ovulate. Each woman is born with a million follicles, the fundamental units of the ovary, each containing a single germ cell (oocyte) surrounded by hormone-producing somatic cells (granulosa and theca). By puberty, that number is reduced by half and by menopause, all of the follicles will have been exhausted. We propose to define the cellular mechanism that initiates growth of a small subset of follicles at a very specific point in time. Our hypothesis is that if we combine powerful new technologies from chemistry, cell biology, physical and computational sciences, we can discover the integrated network of signaling cues required for follicle activation, what we call the ovarian calendar program.
The main goal of this program grant is to investigate signaling pathways by which hormones or other regulatory factors act on the ovary to promote steroidogenesis and the maturation of follicle and germ cells necessary to sustain female fertility.
The goal of the Northwestern University Superfund Research Center in Reproductive Health Hazards (NU SRC) is to focus on the effects of environmental metal contaminants on reproductive function by determine the impact of metals on gamete (egg and sperm) function and reproductive health, developing novel bioassays for assessing the risk of environmental metals on reproductive function and determine the longitudinal risk of environmental metals exposure to human reproductive health. We will coordinate this effort with an evaluation of the routes by which environmental metal contaminants enter human populations in coordination with the development of methods for detecting and removing select metals that pose a threat to human health.
The female reproductive tract is an integrated set of organs that supports women's overall endocrine health, fertility and fetal development. Each organ within the tract is composed of different cells that interact with each other, relying on a precise tissue architecture that is more effectively studied in three-dimensional (3D) tissue cultures. The team’s goal is to develop and validate 3D culture systems of the five major organs of the reproductive tract into an Ex Vivo Female Reproductive Tract Integration In a 3D Microphysiologic System. It can then be used to measure responses to normal hormones, endocrine disruptors, and other reproductive hazards and in the drug development pipeline.
A grim health diagnosis that threatens to damage reproductive health presents both patients and providers with the dual challenges of maximizing life expectancy while protecting the ability to have a family-after the disease. The Northwestern University Center for Reproductive Health After Disease addresses the science needed to understand human follicle and egg biology and pursue cutting-edge options for preserving reproductive health. It also provides physicians, patients, their families, and the public with information about the risks of diseases and treatments available to protect reproductive health.
The most recognized action of activin is to stimulate follistatin-stimulating hormone (FSH) production by the pituitary gonadotrope in vitro and in vivo. Our work in the past five years has led to a consolidated model of activin-controlled pituitary FSH regulation and an expansion of our understanding regarding the species-specified role of activin in the gonadtropin surge spacing.
The objective of this research is to understand the structural determinants that influence inhibin heterodimer assembly, processing and antagonism of an activin signal. The most recognized action of inhibin is to suppress FSH production and secretion by the pituitary gonadotrope in vitro and in vivo.
In 2007, Dr. Teresa Woodruff and colleagues at Northwestern University and the University of Chicago received the prestigious Medical Research Award from the W. M. Keck Foundation.
Every year, more than 135,000 young people are diagnosed with cancer in the US. Seventy-seven percent of cancer patients under age 45 survive more than 5 years and quality-of-life is important to survivors, their clinicians, and their communities.
The Center for Reproductive Research (CRR) at Northwestern University supports a multidisciplinary group of scientists who use innovative approaches to expand the understanding of female fertility and infertility. The purpose of our center is to shed light on the structure-function relationships between cells in the ovary and the hormones that regulate follicle development.



 Repropedia is a reproductive dictionary created by an international team of scientists and clinicians with definitions that are both accurate and accessible to all. By actively participating in Repropedia, the reproductive science community disseminates knowledge and ensures that the next generation of learners has an information resource suited to their learning needs.
Repropedia is a reproductive dictionary created by an international team of scientists and clinicians with definitions that are both accurate and accessible to all. By actively participating in Repropedia, the reproductive science community disseminates knowledge and ensures that the next generation of learners has an information resource suited to their learning needs. 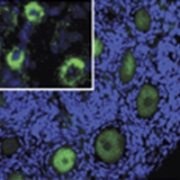 This work identifies the mechanism of the c-Abl kinase inhibitor imatinib mesylate, which has recently been identified as a fertoprotective agent against the alkylating chemotherapy cisplatin. We show that, in vitro, imatinib prevents cisplatin-induced apoptosis of oocytes within primordial follicles by inhibiting c-Able kinase and downstream activation of TAp73-BAX-mediated apoptosis.
This work identifies the mechanism of the c-Abl kinase inhibitor imatinib mesylate, which has recently been identified as a fertoprotective agent against the alkylating chemotherapy cisplatin. We show that, in vitro, imatinib prevents cisplatin-induced apoptosis of oocytes within primordial follicles by inhibiting c-Able kinase and downstream activation of TAp73-BAX-mediated apoptosis.